The analysis of the entire transcriptome by microarrays (or DNA chips) provides the qualitative and quantitative gene expression profile of a sample (cells, tissues, biopsies), at a given moment or in an identified physiological or pathological condition. As this profile is an inherent characteristic of a sample, it can be used for numerous applications: discovery of new therapeutic targets, identification of biomarkers, analysis of mechanisms of action or of signaling pathways, fundamental knowledge, etc.
Bioalternatives, an Affymetrix® service provider, offers transcriptome analysis by microarrays (DNA chip technology), using GeneAtlas®platform.
Microarrays
Bioalternatives has a wide range of Affymetrix microarrays:
- Human Genome U219 Array Strip
- Human Genome U133+ PM Array Strip
- Rate Genome-230 PM Array Strip
- Mouse Genome-430 PM Array Strip
- Human Gene ST Array Strip
- Rate Gene ST Array Strip
- Mouse Gene ST Array Strip
- Canine Gene ST Array Strip
- Feline Gene ST Array Strip
- miRNA Array Strip
In addition to the gene expression analysis for the whole transcriptome, Bioalternatives offers the analysis of microRNA profiles, by using the miRNA 4.1 chip dedicated to the detection of small non-coding RNAs.
Gene Array Strips are also available to perform studies on samples from other organisms. Please do not hesitate to contact us for any additional information.
Microarrays, analyses and associated services
Our microarray analysis services include:
- Biological sample preparation and recovery
- RNA extraction and quality control using electrophoresis (Agilent)
- Hybridization on microarrays
- Scanning
- Raw data at CEL format
- Quality control and data normalization
as well as optional additional services related to bioinformatics analysis and data processing:
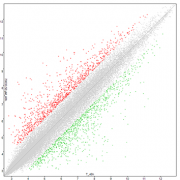
- Standard analysis of data (calculation of fold change and statistical analysis)
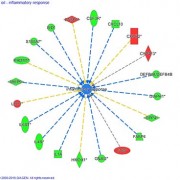
- Basic functional analysis: inter-group comparisons and study of modulated biological process (DAVID, IPA)
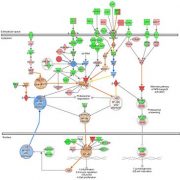
- Advanced functional analysis, including clustering and identification of target-genes and/or modulated signaling pathways (IPA)