Effects on clinical signs, lipids and hydration factors of combined applications of shampoo and mousse containing Ophytrium and Seboliance in seborrheic dogs
J. Kondratjeva1 – J. Brun1 – N. Amalric2 – F. Moog1 – D. Combarros1;3 – C. Pressanti1 – C. Zemirline4 – N. Maubert4 – M. Gatellet4 – M.C. Cadiergues1;3
1Small Animal Clinic, Université de Toulouse, ENVT, Toulouse, France – 2Synelvia, Labège, France – 3UDEAR, Université de Toulouse, INSERM, ENVT, Toulouse, France – 4Ceva Santé Animale, France
DESCRIPTION
Performance of the DOUXO® S3 SEB shampoo and subsequent mousse applications (CEVA, France) was assessed over three weeks in 18 seborrheic dogs in a prospective, controlled study. The protocol combining shampoo and leave-on mousse applications resulted in a significant clinical improvement, the maintenance of skin hydration, initial decrease and further restoration in hair lipids.
INTRODUCTION
Seborrhea is frequent and time-consuming1
Shampoos are often effective2 but
• procedure takes time
• are often hard to realize
• a combination of different types is often necessary
Ophytrium3, a purified natural ingredient, has a high tolerance and a simultaneous threefold action on the skin barrier: it strengthens the mechanical barrier, limits the adhesion and biofilm formation and soothes the skin.
Seboliance4, a natural plant extract, has seboregulating properties.
OBJECTIVE
Evaluate the protocol combining applications of one shampoo and subsequent mousses containing Ophytrium and Seboliance (DOUXO® S3 SEB, Ceva Santé Animale, Libourne, France) in seborrheic dogs on:
• tolerance
• performance: clinical score (CS), pruritus, skin surface cytology, natural moisturizing factors content (NMFs), hair surface lipids content (HL)
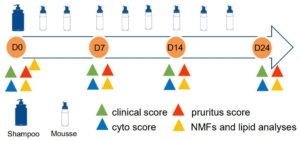
MATERIALS & METHODS
Animals
18 adult dogs (griffons, Bruno du Jura and Bleu de Gascogne)
• idiopathic greasy seborrhea
• same diet/environment prior and during the study
• randomly allocated in placebo (plain water – 6 dogs) or test (12 dogs) group
Study design
REFERENCES
1. JS Yoon at al., Vet Dermatol, 2013, 24:84-89.e21-22.
2. W Rosenkrantz, Clin Tech Small Anim Pract, 2006, 21:106–16.
3. MH Chen et al., J Ethnopharmacol, 2016, 181:193–213.
4. K Barathikannan et al., BMC Complement Altern Med, 2016,16:264.
The study was funded by Ceva.





Performance and Tolerance of a Protocol for Idiopathic Chronic Greasy Seborrhea in 18 Dogs Using a Shampoo and Mousse Containing Plant Extracts
Veterinary Medicine, Veterinary medicineThe study aimed to evaluate the tolerance, performance and effect on hair lipids and skin hydration of a protocol combining applications of one shampoo and subsequent mousses containing plant extracts (Ophytrium and Seboliance) in dogs with an undiagnosed chronic greasy keratinisation disorder.
Clinical and Microbiological Performances and Effects on Lipid and Cytokine Production of a Ceruminolytic Ear Cleaner in Canine Erythemato-Ceruminous Otitis Externa
Veterinary medicine, Veterinary MedicineErythemato-ceruminous otitis externa (ECOE) is the most common type of otitis in dogs and is generally associated with bacterial and/or yeast infections.
Multiplex Cytokine Analyses in Ear Canals of Dogs Suggest Involvement of IL-8 Chemokine in Atopic Otitis and Otodectic Mange—Preliminary Results
Veterinary Medicine, Veterinary medicineAtopic dermatitis is a form of allergy of genetic origin accompanied by itching, during which the animal more easily develops allergies to environmental factors, usually pollen and dust mites.
Survey of cytokines on ocular surfaces of atopic dogs by multiplex analysis using two sampling methods – a pilot Study
Veterinary medicine, Veterinary MedicineConjunctivitis in atopic dogs has already been described yet is rarely observed, and likely underdiagnosed in practice.
Effects on clinical signs, lipids and hydration factors of combined applications of shampoo and mousse containing Ophytrium and Seboliance in seborrheic dogs
Veterinary medicinePerformance of shampoo and subsequent mousse applications was assessed in seborrheic dogs. Shampoo and leave-on mousse applications resulted in a significant improvement…
Evaluation of the anti-inflammatory and immunomodulatory effects of a product using in vitro canine whole blood models
Veterinary medicine2D and 3D in vitro canine models
Veterinary medicineEvaluation of the effect of a reference molecule on an Atopic Reconstructed Canine Epidermis model (RCE-AD)
Veterinary medicineA prospective, randomized, double blind, placebo-controlled evaluation of the effects of an n-3 essential fatty acids supplement (Agepi® ω3) on clinical signs, and fatty acid concentrations in the erythrocyte membrane, hair shafts and skin surface of dogs with poor quality coats
Veterinary medicineRefined Immunochemical Characterization in Healthy Dog Skin of the Epidermal Cornification Proteins, Filaggrin, and Corneodesmosin
Barrière cutanée et hydratation, Skin barrier and hydration, Veterinary medicine, Veterinary MedicineFilaggrin (FLG) and corneodesmosin (CDSN) are two key proteins of the human epidermis.